|
To determine the electron configuration for any particular atom, we can “build” the structures in the order of atomic numbers. The notation 3 d 8 (read "three–d–eight") indicates eight electrons in the d subshell (i.e., l = 2) of the principal shell for which n = 3.įigure 6.25 The diagram of an electron configuration specifies the subshell ( n and l value, with letter symbol) and superscript number of electrons. A superscript number that designates the number of electrons in that particular subshell.įor example, the notation 2 p 4 (read "two–p–four") indicates four electrons in a p subshell ( l = 1) with a principal quantum number ( n) of 2.  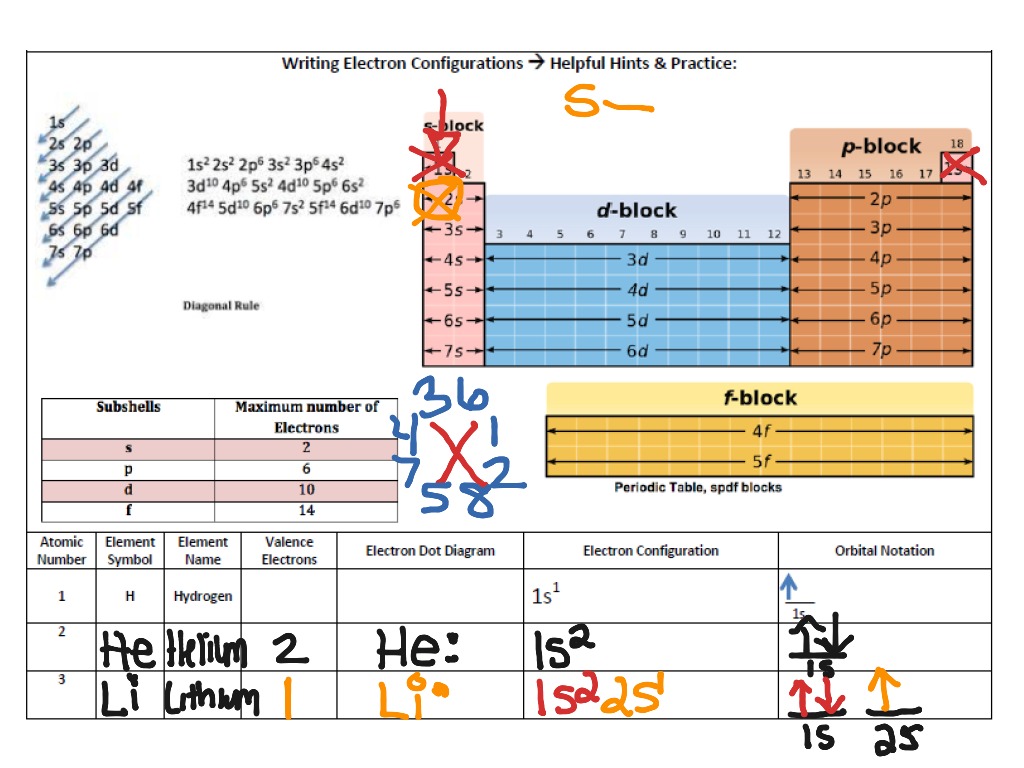
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
